SOT 65th Annual Meeting and ToxExpo 2026
San Diego Convention Center, San Diego, California
March 22, 2026 - March 25, 2026The SOT Annual Meeting and ToxExpo brought together a global community of toxicologists and scientists from related disciplines. Check out our Exhibitor-Hosted Session and poster presentations from the event and browse our resources below to learn about our HepatoXcell™ primary human hepatocytes, peripheral blood mononuclear cells (PBMCs), hTERT-immortalized cells, and primary cells that support predictive drug development and toxicity testing.
Primary human hepatocytes are considered the gold standard for in vitro liver models due to their high predictive value in drug metabolism and toxicity studies. They can provide early insights into how a drug will behave in the human body, potentially reducing the risk of adverse effects in clinical trials. ATCC hepatocytes are meticulously isolated and characterized to ensure the highest quality and performance for your drug development and toxicity testing needs. Try our HepatoXcell™ primary human hepatocytes and media today!
Learn moreCheck out our Exhibitor-Hosted Session
Shaping the Future of Predictive Toxicology with HepatoXcell for Advanced MPS Platforms
Presenter: Carolina Lucchesi, PhD, Principal Scientist, ATCC
Date & time: Tuesday, March 24, 2026, from 10:45–11:45 AM
Location: Room 23C
This session is an Exhibitor-Hosted Session. Although not an official part of the SOT Annual Meeting scientific program, its presentation is permitted by the Society.
Access our Tiny Tox Talk Presentation
Mini Organs, Major Insights—Toxicology Goes 3-D
Presenter: Carolina Lucchesi, PhD, Principal Scientist, ATCC
Date & time: Wednesday, March 25, 2026, from 11:20–11:40 AM
Location: Tiny Tox Talks Theater
Get a copy of our posters
Generation of a Novel Immortalized Human Corneal Epithelial Cell Line for Reliable In Vitro Ocular Toxicity Studies
Presenter: Xiangshan Zhao, PhD, Senior Scientist, ATCC
Date & time: Tuesday, March 24, 2026, from 9:15–11:45 AM
Location: ToxExpo, Hall B
Poster Board Number: M839
Abstract Number: 4340
ATCC’s Human-Relevant Cancer Models for Mechanism-Based Toxicity and Drug Response Profiling
Presenter: Ajeet Singh, PhD, Senior Scientist, ATCC
Date & time: Tuesday, March 24, 2026, from 9:15–11:45 AM
Location: ToxExpo, Hall B
Poster Board Number: H622
Abstract Number: 4140
Human Liver-on-a-Chip Systems for Enhanced Mechanism-Based Toxicity Screening
Presenter: Carolina Lucchesi, PhD, Principal Scientist, ATCC
Date & time: Tuesday, March 24, 2026, from 9:15–11:45 AM
Location: ToxExpo, Hall B
Poster Board Number: H618
Abstract Number: 4136
Learn more about our toxicology portfolio
Primary Human Hepatocytes
Discover how HepatoXcell™ can support your predictive drug development and toxicity testing.
MoreRNA-seq Reveals Comprehensive Gene Expression Landscape in Primary Human Hepatocytes
This application note shows how RNA-seq reveals key gene expression differences in primary human hepatocytes from different donors.
MoreExploring the performance of HepatoXcell™ in a variety of culture formats
In this application note, we demonstrate that the HepatoXcell™ system supports diverse culture formats, offering flexibility for tailoring experimental setups for liver biology research, disease modeling, drug discovery, and toxicity testing.
MorePeripheral Blood Mononuclear Cells
ATCC PBMCs are cryopreserved, ethically sourced immune cells with high viability, ideal for reliable immunology research and functional assays.
More Brochure
Brochure
Toxicology Portfolio
ATCC has the credible models that you need to perform standardized, reliable, and reproducible toxicology studies.
More Culture guide
Culture guide
ATCC Animal Cell Culture Guide
This guide contains general technical information for working with animal cells in culture, including media, subculturing, cryopreservation, and contamination.
More Application note
Application note
Comparing Toxicological Responses Between ALI-Incubated Primary Airway Epithelial Cells and Undifferentiated Counterparts
In this application note, we show that self-constructed airway models may serve as useful tools future airway toxicity research.
More Application note
Application note
Genetically Modified Renal Proximal Tubule Epithelial Cells - A Physiologically Relevant Renal Solute Carrier Uptake Model for Drug Toxicity Studies
We created three different hTERT-immortalized human primary renal proximal tubule epithelial cells (RPTEC) that stably express the OAT1, OCT2, and OAT3 proteins. We then tested the ability of these cells to intracellularly convey known SLC protein substrates.
More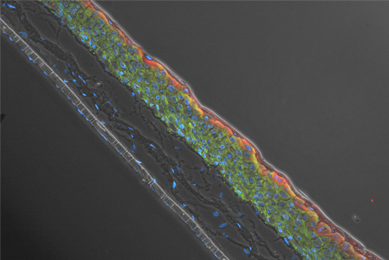 Application note
Application note
hTERT-immortalized and Primary Keratinocytes Differentiate into Epidermal Structures in 3D Organotypic Culture
In this study, we confirmed that an immortalized keratinocyte cell line, Ker-CT, is able to differentiate into organotypic skin equivalents in a 3D culture model
More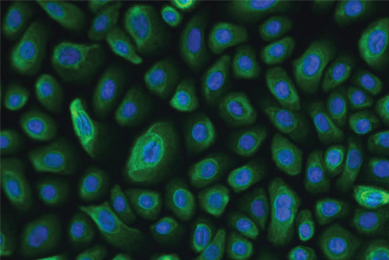 Brochure
Brochure
hTERT-immortalized Primary Cells
Enjoy the best of all worlds with human telomerase reverse transcriptase (hTERT)-immortalized Primary Cells from ATCC.
More Brochure
Brochure
Primary Cell Solutions
ATCC Primary Cell Solutions is a system of matched components designed to boost growth, maximize functionality, and maintain normal morphology for each primary cell type.
MorePrimary Human Immune Cells
ATCC primary immunology cells are able to support complex, physiologically relevant research projects, including toxicity screening, transplantation and graft rejection, inflammation and allergy, vaccine, drug development, and cancer immunology studies.
MoreContact us today
Your ATCC sales representative stands ready to discuss your research needs and provide you with personalized care. Please complete our form to let us know how we can help, and we will start working on your needs right away.