Dr. Shapiro: Good afternoon, Elisia and Birgitt, thank you for meeting with me today to discuss The Michael J. Fox Foundation for Parkinson’s Research (MJFF) and the cell models that were made available through a collaboration between MJFF and ATCC. Elisia, can you tell me a bit about the foundation?
Dr. Clark: The Michael J. Fox Foundation for Parkinson’s Research (MJFF) was founded in 2000 by the actor Michael J. Fox and is dedicated to finding a cure for Parkinson's disease through an aggressively funded research agenda and by ensuring the development of improved therapies for those living with Parkinson's today. This is achieved by identifying and funding projects most vital to patients and spearheading solutions to address field-wide challenges. To date, MJFF has put more than $1 billion in research program funding and made 200 preclinical tools available.
Dr. Shapiro: How does the collaboration between MJFF and ATCC help researchers?
Dr. Clark: MJFF has a Tools Program that seeks to de-risk the Parkinson’s disease research field and speed progress towards a cure by developing and distributing well-validated, high-quality preclinical research tools that fill field-wide needs. MJFF has two methods for making preclinical tools available, one is the Tool Generation Program in which MJFF generates novel tools, such as the LRRK2 mutant MEF lines available through ATCC. The other is the MJFF Sponsored Tools Program, which identifies preclinical tools developed by external research investigators, such as the patient-derived iPSC lines generated by Dr. Birgitt Schüle, and connects them with repositories, such as ATCC, for widespread distribution.
Dr. Shapiro: Will there be other MJFF cell lines developed from different individuals?
Dr. Clark: Yes, we have multiple ongoing efforts to generate patient-derived iPSC lines. Notably, various patient-derived iPSCs were generated within a sub-study of our landmark Parkinson’s Progression Markers Initiative (PPMI) study. These lines represent various cohorts including healthy controls, prodromal and idiopathic Parkinson’s disease, as well as lines from patients that harbor GBA, LRRK2, and/or SNCA mutations with and without Parkinson’s disease, with mutation-corrected isogenic controls soon to become available.
Dr. Shapiro: Birgitt, can you tell us how the collaboration between MJFF and ATCC has affected your research?
Dr. Schüle: The MJFF has dedicated significant efforts over the past few years to create a range of exceptional, well-characterized, and cost-effective research tools. These tools encompass antibodies, cell lines, and transgenic animal models, among others. These resources play a pivotal role for researchers, myself included, by providing access to shared research reagents and tools. This not only enhances the reproducibility of data but also allows for comparability across different studies. The outcome of this collaborative approach expedites the pace of discovery that ultimately leads to new treatments for individuals with Parkinson’s disease.
Dr. Shapiro: There have been several Synuclein Alpha (SNCA) knockout isogenic cell lines that were made available through the collaboration between MJFF and ATCC. What is the significance of these cell lines?
Dr. Schüle: The cell lines were derived from a patient with an SNCA genomic triplication, who was a descendent of the "Iowa kindred," a multi-generational family hailing from Iowa. This family has been under clinical observation at the Mayo Clinic since the 1920s due to their inherited form of autosomal-dominant Parkinson's disease.
First documented by Spellman in 1962, the Iowa kindred displays a distinct pattern of severe parkinsonism that follows an autosomal-dominant inheritance pattern. Members of this family, with ancestral roots in both English and German heritage, exhibit an unusually early onset of the disease, with symptoms emerging at an average age of 34 years, within a range of 20 to 48 years. The disease progression within this kindred is fast, with affected individuals succumbing to dementia and passing away within 2 to 12 years of symptom onset.
Postmortem analysis of affected individuals has unveiled significant degeneration of the substantia nigra, extensive presence of Lewy bodies in both subcortical and cortical regions, cortical vacuolation, loss of nerve cells, and gliosis in the hippocampus. Alpha-synuclein staining revealed neuritic pathology in cortical regions that surpassed the severity observed in even the most pronounced cases of dementia with Lewy bodies.
In 2003, Singleton and collaborators1 identified in the Iowa kindred a triplication of the SNCA genomic locus located on chromosome 4q21. Initially designated as PARK4, the size of this triplication is 1.7 Mb, encompassing a total of 17 genes. The triplication specifically affected the SNCA gene on one allele, which results in a twofold increase in alpha-synuclein protein expression within the brain.
This groundbreaking discovery underscores the pivotal role of an elevated expression of wild-type alpha-synuclein in triggering a neurodegenerative condition strikingly reminiscent of Parkinson's disease. Dr. James William “Bill” Langston, a notable mentor in this field, coined these cases of SNCA triplication as "Parkinson's on steroids," given the accelerated disease trajectory and pronounced neuropathological changes observed in affected individuals.
Stay tuned for the second interview in this series where Dr. Schüle will detail the development and characterization of CRISPR genome-edited SNCA knockout induced pluripotent stem cells and describe their potential uses in Parkinson’s disease research.
Did you know?
ATCC offers wild-type LRRK2, LRRK2 knockout, and human LRRK2 T1348N (GTPase-dead) knock-in macrophage cell lines for Parkinson’s disease research.
Meet the scientists
Elisia Clark, PhD
Associate Director of Research Programs, The Michael J. Fox Foundation for Parkinson’s Research
Dr. Elisia Clark is an Associate Director of Research Programs at the Michael J. Fox Foundation for Parkinson’s disease. Her scientific research interests began as a Meyerhoff and Howard Hughes Medical Institute Scholar at University of Maryland, Baltimore County. She then received her PhD in Pharmacology from the University of Pennsylvania researching molecular mechanisms of neurodegenerative diseases. Now, as a Scientist at The Michael J. Fox Foundation, she is making an impact beyond the bench to support research that accelerates meaningful therapeutic advancements towards a cure for Parkinson’s disease through the Michael J. Fox Foundation Sponsored Tools Program which identifies and distributes preclinical tools generated by external research investigators to fill gaps needed by the research community.
Birgitt Schüle, MD
Associate Professor, Department of Pathology, Stanford University School of Medicine
Birgitt Schüle, MD, is an Associate Professor in the Department of Pathology at Stanford University School of Medicine. Her research focuses on medical genetics and stem cell modeling to uncover disease mechanisms and pathways involved in neurodegeneration in Parkinson's disease and related disorders. She is dedicated to developing novel therapeutic strategies that contribute to the advancement of precision medicine.
Dr. Schüle received her medical training from the Georg-August University Göttingen and Medical University Lübeck, Germany, during between 1993 and 2001. In 2001, she obtained her doctoral degree in medicine (Dr. med.) from the Georg-August University Göttingen in neurophysiology. Following, from 2001 to 2002, she completed her neurology internship at the Medical University of Lübeck, where she was mentored by Prof. Christine Klein in neurogenetics of Parkinson’s disease and dystonia syndromes. Continuing, Dr. Schüle pursued a postdoctoral fellowship in human genetics at Stanford University School of Medicine from 2003 to 2005, under the guidance of Prof. Uta Francke.
Between 2005 and 2019, Dr. Schüle built and led critical clinical research programs and establishing biospecimen and cell line repositories in the fields of neurogenetics, translational stem cell research, and brain donation at the Parkinson's Institute and Clinical Center.
Dr. Schüle serves as Associate Core Leader, Neuropathology, within the Stanford Alzheimer Research Center (ADRC). Within this role, she has made significant contributions to the ADRC, particularly in genetic characterization, biobanking, and the establishment of a human induced pluripotent stem cell and post-mortem leptomeninges tissue bank. These resources are made accessible to the scientific community through repositories at the National Institutes of Health (NIH), thereby promoting collaborative research.
Dr. Schüle's expertise in the field of neurodegeneration have been instrumental in advancing medical knowledge. She is highly regarded within the scientific community for her significant contributions and plays an essential role in the pursuit of effective treatments and precision medicine approaches for neurodegenerative conditions.
Brian Shapiro, PhD
Marketing Segment Manager, Oncology, ATCC
Brian A Shapiro, PhD, works to communicate the scientific breakthroughs of ATCC’s product development laboratories to the biomedical research community. Brian is the Executive Producer of ATCC's Podcast, Behind the Biology. Previously, he worked at Virginia Commonwealth University, where he investigated the role of pre-mRNA splicing in the multi-drug resistance of lung cancer. Dr. Shapiro attended the Medical College of Georgia, where his research focused on adrenal physiology as well as diseases of the epidermis.
Explore our featured resources
MJFF Cell Lines
ATCC has been selected by The Michael J. Fox Foundation for Parkinson’s Research (MJFF) to provide vital cell lines to academic, pharmaceutical, and biotechnology organizations committed to speed a cure for Parkinson’s disease.
More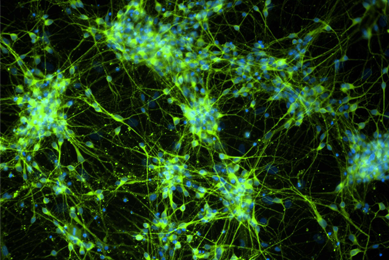
Neuroscience
At ATCC, we support neuroscience research with authenticated normal and disease cells and biomaterials you can rely on.
More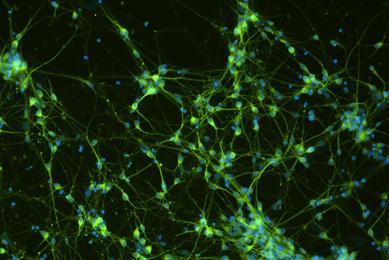
Neural Progenitor Cells
ATCC cells are valuable for investigators looking to reduce the time from initial iPSC culture to experiment readout, as they eliminate the 4 to 8 weeks for iPSCs to differentiate into NPCs.
MoreReferences
- Singleton A.B., et al. Alpha-synuclein locus triplication causes Parkinson’s disease. Science 302, 841 (2003).
- Zafar, F. et al. Genetic fine-mapping of the Iowan SNCA gene triplication in a patient with Parkinson's disease. NPJ Parkinsons Dis 4, 18 (2018).
- Mittal, S. et al. beta2-Adrenoreceptor is a regulator of the alpha-synuclein gene driving risk of Parkinson's disease. Science 357, 891-898 (2017).
- Oliveira, L.M. et al. Elevated alpha-synuclein caused by SNCA gene triplication impairs neuronal differentiation and maturation in Parkinson's patient-derived induced pluripotent stem cells. Cell Death Dis 6, e1994 (2015).
- Flierl, A. et al. Higher vulnerability and stress sensitivity of neuronal precursor cells carrying an alpha-synuclein gene triplication. PLoS One 9, e112413 (2014).
- Chung, C.Y. et al. Identification and rescue of alpha-synuclein toxicity in Parkinson patient-derived neurons. Science 342, 983-7 (2013).
- Mak, S.K., Tewari, D., Tetrud, J.W., Langston, J.W. & Schüle, B. Mitochondrial Dysfunction in Skin Fibroblasts from a Parkinson’s Disease Patient with an alpha-Synuclein Triplication. Journal of Parkinson's disease 1, 175-183 (2011).
- Byers, B. et al. SNCA triplication Parkinson's patient's iPSC-derived DA neurons accumulate alpha-synuclein and are susceptible to oxidative stress. PLoS One 6, e26159 (2011).
- Zafar, F., Nallur Srinivasaraghavan, V., Yang Chen, M., Alejandra Morato Torres, C. & Schüle, B. Isogenic human SNCA gene dosage induced pluripotent stem cells to model Parkinson's disease. Stem Cell Res 60, 102733 (2022).