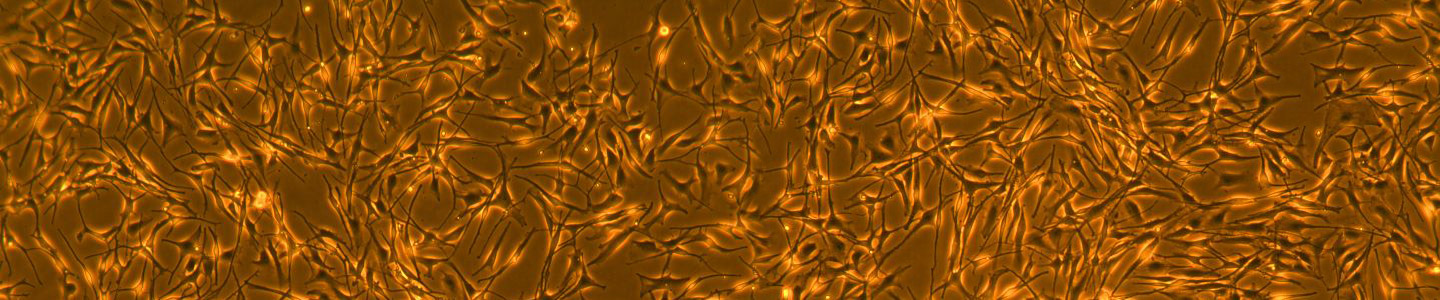
Development of hTERT-Immortalized Neonatal Melanocytes for Toxicity Studies and Melanogenesis Regulation
ACT 2022 Annual Meeting
Denver, Colorado, United States
November 14, 2022Abstract
Download the poster to explore the use of hTERT-immortalized melanocytes as in vitro cell models for skin toxicology studies
DownloadWatch the poster presentation
Presenters
Diana Douglas, BS
Lead Biologist, ATCC
Diana Douglas is a Lead Biologist at ATCC. For the last nine years, she has focused her research on the development of advanced biological models including the use of CRISPR/Cas9 gene-editing technology. Previously, Ms. Douglas worked at the Baker Institute for Animal Health at Cornell University and the Dalton Cardiovascular Research Center at the University of Missouri, where her research focused on the mechanisms of necrotic cell death in heart disease. Ms. Douglas attended Truman State University where she obtained a Bachelor of Science in Biology.
Steven Budd, MS, MBA
Product Specialist, ATCC
Steven Budd is a Product Specialist that manages the cell culture reagents at ATCC. He has 6 years of experience in the product management of scientific tools. Before that, he gained 4 years of experience in biomedical research and cell culture as a research specialist at the University of North Carolina at Chapel Hill. Mr. Budd has a M.S. in Biology from the University of North Carolina at Wilmington and an M.B.A. from North Carolina State University.
Toxicology testing products
ATCC knows that worthwhile science takes time, especially in the toxicology, pre-clinical stages of drug development. It is critical that the standards and model organisms used in toxicological testing are reliable and authenticated. We can help streamline your research by providing the most authenticated, advanced, and functional models available. Let ATCC revolutionize and accelerate your toxicology studies in every phase of the research and testing process.
ATCC provides the cells, media, and reagents needed to explore each step of the in vitro preclinical testing process—from modeling, screening, and characterization to exploratory toxicology to pharmacokinetics and metabolism. We provide renal, neural, airway, and skin models for such applications as high-content screening, 3D culture, spheroid culture, permeability assays, metabolic stability and survival studies, transport activity measurement, and more.
Explore toxicology tools