An In Vitro Cell Model for Toxicity Studies and Pigmentation Regulation Utilizing hTERT-immortalized Neonatal Dermal Melanocytes
SOT 59th Annual Meeting and ToxExpo 2020
Anaheim, California, United States
March 15, 2020Abstract
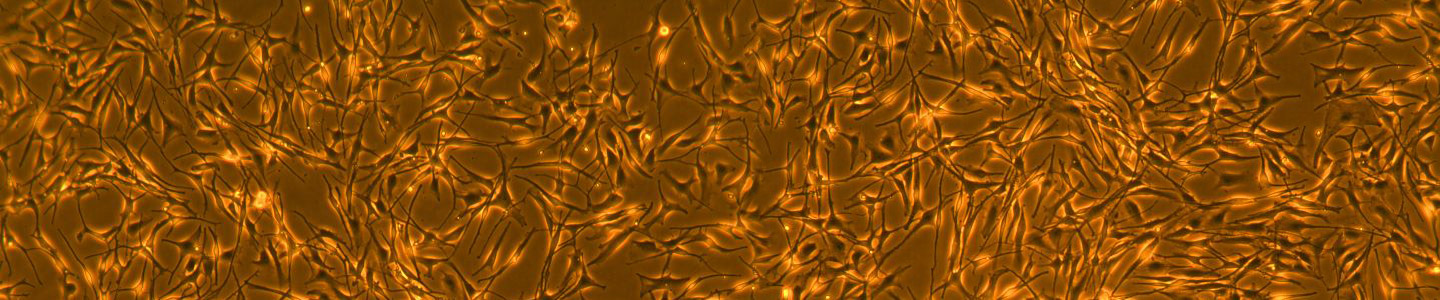
Skin pigmentation is a complex process; melanocytes produce melanin and package it into melanosomes that are in turn exocytosed, then endocytosed by neighboring keratinocytes. Numerous genes play roles in controlling pigmentation at various levels of melanin production. Mutations in these genes are characteristic of multiple skin disorders, including hyperpigmentation, hypopigmentation, and mixed hyper-/hypopigmentation. Additionally, extrinsic factors secreted by the surrounding resident cell types also regulate the melanin expression in melanocytes. Human primary cells can be a useful model for elucidating melanocyte biology and their toxicity to dermal agents. However, primary cells have their limitations such as donor variability and limited lifespan. Consequently, a need exists for a more robust human cell model system for the study of skin pigmentation and agents which damage melanocytes.
In this study, we immortalized primary epidermal neonatal melanocytes by expressing human telomerase reverse transcriptase (hTERT) in cells that were isolated from a neonatal donor. The immortalized primary melanocytes were cultured continuously for more than 35 population doublings without any signs of replicative senescence, yet retained melanin production. The immortalized primary melanocytes maintained a consistent expression of the melanocyte-specific marker TRP-1, and lacked expression of the fibroblast-specific marker TE7. In addition, we demonstrate the capability of these immortalized primary neonatal melanocytes to transfer melanosomes to keratinocytes utilizing a 3-D human dermal organotypic culture, and the ability to modulate melanogenesis with depigmentation agents and these agents’ effects on cell viability. Taken together, the hTERT immortalized primary neonatal melanocytes described here provide a versatile in vitro cell model for the study of toxicity and melanogenesis regulation.
Watch the presentation
Toxicology testing products
ATCC knows that worthwhile science takes time, especially in the toxicology, pre-clinical stages of drug development. It is critical that the standards and model organisms used in toxicological testing are reliable and authenticated. We can help streamline your research by providing the most authenticated, advanced, and functional models available. Let ATCC revolutionize and accelerate your toxicology studies in every phase of the research and testing process.
ATCC provides the cells, media, and reagents needed to explore each step of the in vitro preclinical testing process—from modeling, screening, and characterization to exploratory toxicology to pharmacokinetics and metabolism. We provide renal, neural, airway, and skin toxicity for such applications as high-content screening, 3D culture, spheroid culture, permeability assays, metabolic stability and survival studies, transport activity measurement, and more.
Explore toxicology tools