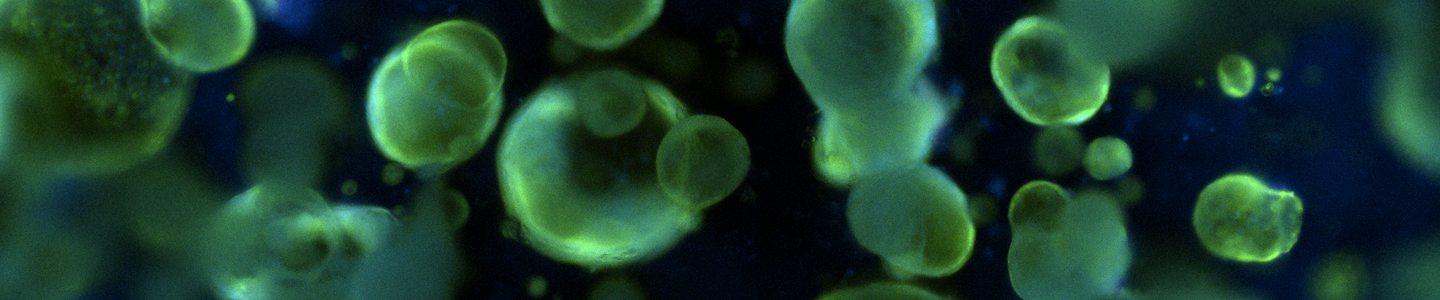
Propagation of Human Colon, Mammary, and Lung Cancer Organoids in Growth Medium Utilizing Tissue-specific Reagent Kits and Ready-to-use Wnt-3a and R-Spondin1 Conditioned Media
Poster
American Association for Cancer Research® (AACR) Annual Meeting 2019
Atlanta, Georgia, United States
March 30, 2019
Abstract
Three-dimensional “organoid” growth of tumors may represent a more physiologically relevant in vitro model system than traditional two-dimensional monolayer cultures of cancer cell lines. With the increased availability of cryopreserved human cancer organoids generated by academic laboratories, large-scale biobanking initiatives, and commercial sources, there is an unmet need for simplified, standardized, and cost-effective methods for preparation of the complex growth media required by these models. Human organoid culture media contains a variety of recombinant proteins, small molecules, and other growth factors that are costly to purchase in small-scale, time consuming to reconstitute and aliquot, and demonstrate varying stability and shelf life once prepared. Organoid culture media often also utilizes undefined conditioned media (CM) from one or more engineered cell lines that must be cultured separately, requiring additional time and resources to maintain. These lines secrete critical growth factors and the CM generated must be carefully prepared, collected, and stored. CM is subject to variability in activity levels due to batch-to-batch and protocol-to-protocol differences that can affect subsequent organoid culture performance. To address these challenges and to facilitate the wide-spread adoption of human organoid culture we are developing reagent kits containing essential growth medium components in an individually lyophilized format for long-term storage and easy single-use preparation of organoid growth media for a variety of tissue types including human colon, mammary, and lung. We compared growth medium formulated with our kit components, with small-scale “homebrew” preparations and found equivalent or better culture performance (as calculated by doubling times) and similar morphology. Both approaches were able to generate media capable of supporting growth for at least three passages (~30 days) in 15 different organoid models. Additionally, we compared protocols and cell lines for CM generation to support organoid culture and found that an optimized, scaled-up production method could generate Wnt-3a and R-spondin1 CM with higher activity levels (up to 52%) and reduced lot-lot variability in comparison with commonly used small-scale protocols. We found that the optimized protocols produced CM that was stable for at least 2 months at 4°C and at least 6 months at -20°C, and was resilient against freezing and thawing. Previously frozen CM was also able to support organoid growth in culture. These results show that our tissue-specific kits and ready-to-use CM can provide a simplified, cost effective method to support the long-term propagation of human cancer organoids from multiple tissues and cancer types.
Download the poster to explore the use of tissue-specific kits and culture media in organoid propagation.
Download
Featured Products
Cell Basement Membrane
Promotes the attachment of hiPSCs and maintains a pluripotent state.
Get the Gel
ROCK Inhibitor Y27632
Increases organoid formation efficiency and recovery from thawing and passaging.
Discover more
L Wnt-3A
Essential for the production of Wnt-3A conditioned medium.
Discover more