What is cell and gene therapy?
Cell therapy aims to treat diseases by restoring or altering certain sets of cells or by using cells to carry a therapy through the body. With cell therapy, cells are cultivated or modified outside the body before being injected into the patient.2 The cells may originate from the patient (autologous cells) or a donor (allogeneic cells).2
As for gene therapy, it is a medical approach used to treat or prevent a genetic disease by adding a new gene or replacing a faulty gene inside the body. To effectively deliver these therapies, engineered viral vectors are often used due to their natural ability to insert genetic material into host cells. Stable cell lines are required for the scalable manufacture of these viral vectors.
Cell and gene therapies have already shown remarkable promise in the treatment of various diseases like cancer, sickle cell disease, retinal dystrophy, beta-thalassemia, and spinal muscular atrophy. The number of cell and gene therapy products on the market and in development will continue to grow as approvals of these treatments quicken. However, despite the success of these treatments, the potential for microbial contamination remains a challenge. Given the short shelf life of batches and urgent patient need, rapid detection of contaminants is essential.
Mycoplasma contamination is a prominent challenge
Mycoplasma is a common cell culture contaminant that poses a major challenge in the manufacture of cell and gene therapies. Mycoplasma contamination results in the loss of an entire product batch and halts production in the manufacturing plant. International regulatory authorities have published guidelines to demonstrate that biological products intended for preventive and therapeutic use and prepared in cell culture substrates must be free of mycoplasma to ensure product safety, purity, and potency. Clean cells are a must.
How we can help
Build your own assay
One option is to use quantitative mycoplasma DNA certified reference materials, which are derived from the strains represented in the ATCC Titered Mycoplasma Reference Strains Panel (ATCC MP-7) and referenced in USP <63>, EP 2.6.7, and JP 18th edition. These DNA certified reference materials are produced under an ISO 17034–accredited process and can be used as controls to establish sensitivity, linearity, and specificity during assay validation or implementation; challenge assay performance; validate or compare testing methods; or benchmark critical assay performance.
Try our detection kit
Our Universal Mycoplasma Detection Kit (ATCC 30-1012K) can be used to detect over 60 species of Mycoplasma, Acholeplasma, Spiroplasma, and Ureaplasma, including the top 8 species most likely to affect cell cultures: M. arginine, M. fermentans, M. hominis, M. hyorhinis, M. orale, M. pirum, M. salivarium, and A. laidlawii. The kit is also able to detect the mycoplasma strains referenced by the pharmacopoeia.
Send us your samples
Our PCR-based mycoplasma testing service is easy to use and fast. You simply order the service, and we’ll send you a sample collection kit, which you can use to spot your cells and then return to ATCC for analysis with results in 3 to 5 business days.
Did you know?
ATCC provides a variety of reporter-labeled cells than can be used to test out CAR-T cell therapies.
Meet the author
Catherine Fox, BS, MBA
Segment Marketing Specialist, ATCC
Catherine works to enable the go-to-market strategy for ATCC products and services that can help researchers working in biopharmaceutical and drug discovery spaces. She works collaboratively with scientists to translate unmet needs to find better solutions. Catherine has a BS in Biology with a minor in Chemistry from Virginia Commonwealth University and received her MBA from George Mason University.
Explore our featured resources
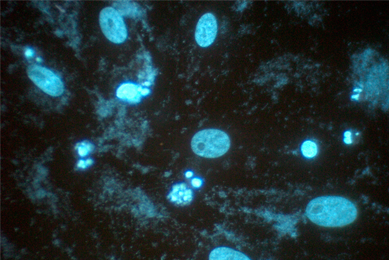
Mycoplasma Testing
Mycoplasma contamination undermines your experimental results and discredits preclinical studies. Your research is too important to risk. Be a part of the movement to raise credibility in science—test your cells for mycoplasma today.
More
Mycoplasma Testing
ATCC’s mycoplasma PCR-based testing service detects over 60 species of Mycoplasma, Acholeplasma, Spiroplasma, and Ureaplasma, with results in 3-5 days. It uses Whatman™ FTA™ sample collection cards with ATCC's Universal Mycoplasma Detection Kit.
More
Mycoplasma Contamination
Mycoplasmas frequently contaminate cell cultures. Discover how rapid and sensitive detection can prevent the costly effects of mycoplasma on your research projects.
MoreReferences
- Mulero A. (2023, June 12). FDA braces for looming boom in cell and gene therapy submissions. BioSpace. https://www.biospace.com/article/fda-braces-for-looming-boom-in-cell-and-gene-therapy-submissions/
- American Society of Gene and Cell Therapy. Gene & Cell Therapy FAQs. (2021). Available online at: https://asgct.org/education/more-resources/gene-and-cell-therapy-faqs (accessed June 29, 2023).