Feasibility of Manufacturing-scale Bioproduction of Novel Next-generation 3-D Organoid Cancer Models in Support of the Human Cancer Models Initiative
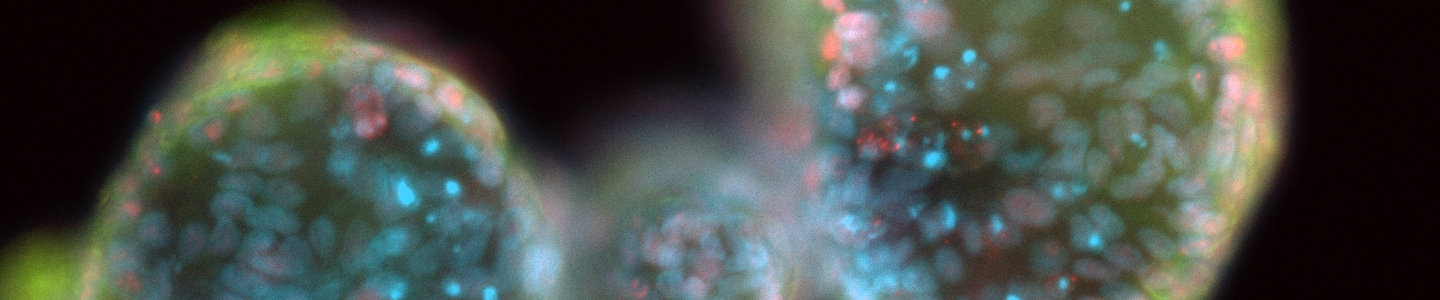
American Association for Cancer Research® (AACR) Annual Meeting 2018
Chicago, Illinois, United States
April 14, 2018Abstract
To meet the need for improved approaches to study cancer in vitro, there has been a surge in the development of novel research models utilizing advanced culture methods. These methods permit in vitro growth of cancer types previously not possible, and/or models with enhanced in vivo relevancy compared to traditional continuous cell lines. However, availability of these early-stage research models is currently limited and there is a lack of data on the ability to scale up production of these models to support the needs of the global research community.
We sought to investigate the protocols, expansion capacity, cryopreservation ability, genetic stability, and feasibility of larger-scale bioproduction of a subset of the models generated by the Human Cancer Models Initiative (HCMI), an international collaborative effort between Cancer Research UK, the foundation Hubrecht Organoid Technology, the National Cancer Institute, and the Wellcome Sanger Institute. The HCMI’s initial goal is the development of 1,000 novel human cancer models, paired with bioinformatics and patient clinical data, particularly from rare or underrepresented cancer types. One advanced culture method being utilized, three-dimensional organoid “microtissue” culture, potentially poses challenges for traditional large-scale bioproduction processes. It requires growth embedded within an undefined extracellular matrix and complex media formulations containing multiple small molecules and recombinant proteins with unknown stability and shelf-life. Additionally, organoid growth media typically includes multiple sources of undefined conditioned media containing critical growth factors.
We cultured organoid models derived from human colon, pancreas, esophagus, and mammary tissues developed by laboratories contributing to the HCMI. Multiple unique donors were available for all tissues and both cancer and non-cancer models were available for two tissue types. Most models were maintained in culture continuously for at least 60 days (7-27 population doublings, > 10 passages). Tissue and donor variability was evident in model characteristics, including morphology (assessed by microscopy and immunocytochemistry), growth rate, and genetic stability (measured by short tandem repeats analysis). All models were amenable to scale up beyond multiwell plates, and all models could recover from cryopreservation.
While organoid culture represents a significant divergence from typical two-dimensional monolayer culture of continuous cell lines, our results show that these next-generation in vitro models are suitable for larger-scale bioproduction. This is vital to ensure the widespread availability of these models within the research community to facilitate applications like pre-clinical drug discovery and basic cancer research.
Download the poster to explore the use of organoids in pre-clinical drug discovery and basic cancer research.
Download